No Cancer Left Behind: Professor’s Imaging Techniques Finds the Less-Visible Cancerous Cells
Kenneth Tichauer, associate professor of biomedical engineering at Illinois Institute of Technology, is developing pioneering cancer imaging techniques to allow surgeons to see cancer cells in the operating room, helping them remove as much cancerous material as possible during surgery while leaving more healthy tissue intact.
Among patients with head and neck cancers, those with some cancer left behind after surgery are 90 percent more likely to die from tumor-related causes within five years than those where all cancer was removed, studies have shown. These scenarios constitute Tichauer’s current focus.
Currently, surgeons often resort to removing extra tissue around the tumor to ensure that all cancer cells are captured. While effective at saving the patient from cancer, taking extra tissue in this region may impact the patient’s ability to eat, breathe, or see.
Tichauer is working with Max Witjes, a professor of head and neck oncology at the University Medical Center Groningen in the Netherlands, who has figured out how to take a method called fluorescence guidance into surgical practice, giving him a closer look at the cancer during an operation.
One way to do this is to inject patients with a dose of fluorescent molecules prior to their surgery. The molecules are designed to attach to epidermal growth factor receptors (EGFR)—proteins that are much more prevalent in tumor cells than healthy cells in adults, so more fluorescent molecules accumulate in the location of the tumor.
“The idea is that when they’re doing their surgery, they can flick the lights off, turn on the fluorescent imaging, and see in their surgical bed if there are any remaining tumor cells,” says Tichauer.
However, at the moment, this method has two main problems.
While highly effective at identifying large groups of cancer cells, the fluorescent molecule will sometimes also attach to healthy cells, leaving the image with a fuzzy background that interferes with the ability to identify small groups of cancer cells.
“When you're doing surgery, you don't care about the big cancer—you can see that by eye—you care about the small, little pieces that are left behind,” says Tichauer.
The second problem is that the current image processing technique used for this type of surgery only shows surgeons cancer cells up to one millimeter below the surface, whereas to get the full picture, they need around five millimeters depth.
Fortunately, Tichauer has the background to tackle both problems through his innovative work with paired-agent imaging and image reconstruction.
“We've been establishing methods to more accurately, non-invasively measure the molecular characteristics of cells, but particularly cancer cells in living patients, for many years,” says Tichauer.
Image fuzziness has been an issue for detecting cancer using fluorescent imaging both in and out of surgery, and in his postdoctoral studies, Tichauer explored ideas for increasing sensitivity.
Since the image fuzziness is caused by the fluorescent molecules attaching to non-cancer cells, Tichauer decided to see if he could measure where that happens and then remove it from the main image.
He did this by introducing a second fluorescent molecule, which emits a different color of light but which has no preference for attaching to cancer cells.
Then he creates a paired-agent image by introducing both fluorescent molecules, taking a photo of the distribution of each color of light in a region, mapping the two images onto each other, and then comparing their intensity.
While small regions of cancer cells blend into the background of a single-agent image—with that background removed, they become visible in the paired-agent image.
“This ratio of the two imaging agents allows us to get a lot more specific information about what is happening in terms of binding,” says Tichauer.
Tichauer’s early studies with paired-agent imaging could distinguish as few as 200 cancer cells in preclinical studies.
In October 2021 Tichauer and his collaborators published a paper in Molecular Imaging and Biology describing how this could be utilized in fluorescent-guided surgery. The preclinical study looked at assessment strategies that could occur during an operation, and it found paired-agent imaging provided a significantly improved ability to distinguish between cancerous and healthy tissue compared to single-agent imaging.
“The aim is giving this cellular sensitivity to surgeons to determine whether or not where they've cut their surgical margin is clear of cancer,” says Tichauer.
They found paired-agent imaging may be able to help surgeons identify micrometastases, or regions with a small number of cancer cells. They also found that this technique was similarly effective across many different samples, unlike single-agent imaging, which is known to work better for some patients than others. Currently, only one fluorescent-imaging agent is approved for clinical use in the United States. With the success of preclinical trials, Tichauer and his collaborators have been working toward getting a second agent approved.
But Tichauer’s major impact has come from combining this innovative imaging technique with innovative methods of processing and interpreting the resulting image, including achieving three-dimensional images.
He received a NSF CAREER award in 2017 and the grand prize in Illinois Tech’s inaugural $1 million Nayar Prize competition with Jovan Brankov, professor of electrical and computer engineering and professor of biomedical engineering at Illinois Tech, in 2019 for their work developing these techniques to improve breast cancer diagnosis.
Now Tichauer has developed an apparatus that brings three-dimensional imaging into the operating room, aiming to help surgeons spot cancer deeper into the tissue. Through Pritzker Institute of Biomedical Science and Engineering’s Exploratory Initiative Program, Tichauer also received funding to work with four biomedical engineering students to carry out the design and construction of the apparatus as part of their spring 2021 senior design course. One of these students, along with Tichauer and a graduate student in his lab, traveled to the Netherlands in summer 2021 to set up the device for preliminary testing by Witjes.
“It’s very similar to how people do image reconstruction with a CAT scan, X-ray, or any other standard 3D clinical imaging,” says Tichauer. But there’s one major difference he had to overcome.
An X-ray image is created by shooting X-rays at the body part and then having a doctor on the other side of the body detect any X-rays that make it through the body. X-rays tend to get absorbed in some parts of the body more than others, so the image created is a straightforward measurement of which X-rays made it through the body and which X-rays got blocked.
But the energy of light used in fluorescent imaging is much lower, and this lower-energy light interacts differently with tissue. Instead of getting absorbed, it tends to scatter.
“It's exactly analogous to driving along in a fog that scatters all the light. It makes it really difficult to see distances and make out where things are. But if your brain actually had a method to model scattering in the new environment, you could see much farther into the fog,” says Tichauer. “So what we do is, we take those images, and we incorporate scattering in the reconstruction model.”
His prototype device incorporates this scattering and combines information from images of the tissue taken at multiple apertures, to look deeper into the tissue, aiming for the five millimeters critical for surgery.
“We innovate in paired-agent imaging methods, and we innovate in the image reconstruction as well. They're distinct, but often we try to bring them together,” says Tichauer.
Tichauer says the long-term goal of his work is personalized medicine.
Every patient’s cancer is unique, and Tichauer says the imaging techniques he’s developing will eventually give doctors enough information to offer much more specific cancer diagnoses and correspondingly personalized treatments. He hopes to reduce the number of surgeries patients are subjected to and improve the outcomes of surgeries when they are unavoidable.
“Biopsy doesn't tell you the whole story of the whole cancer. If we can do it through imaging, then we'll get more information about the entirety of the cancer inside a patient and be able to tune the therapy accordingly,” says Tichauer. “I think we might get there in 20 years.”
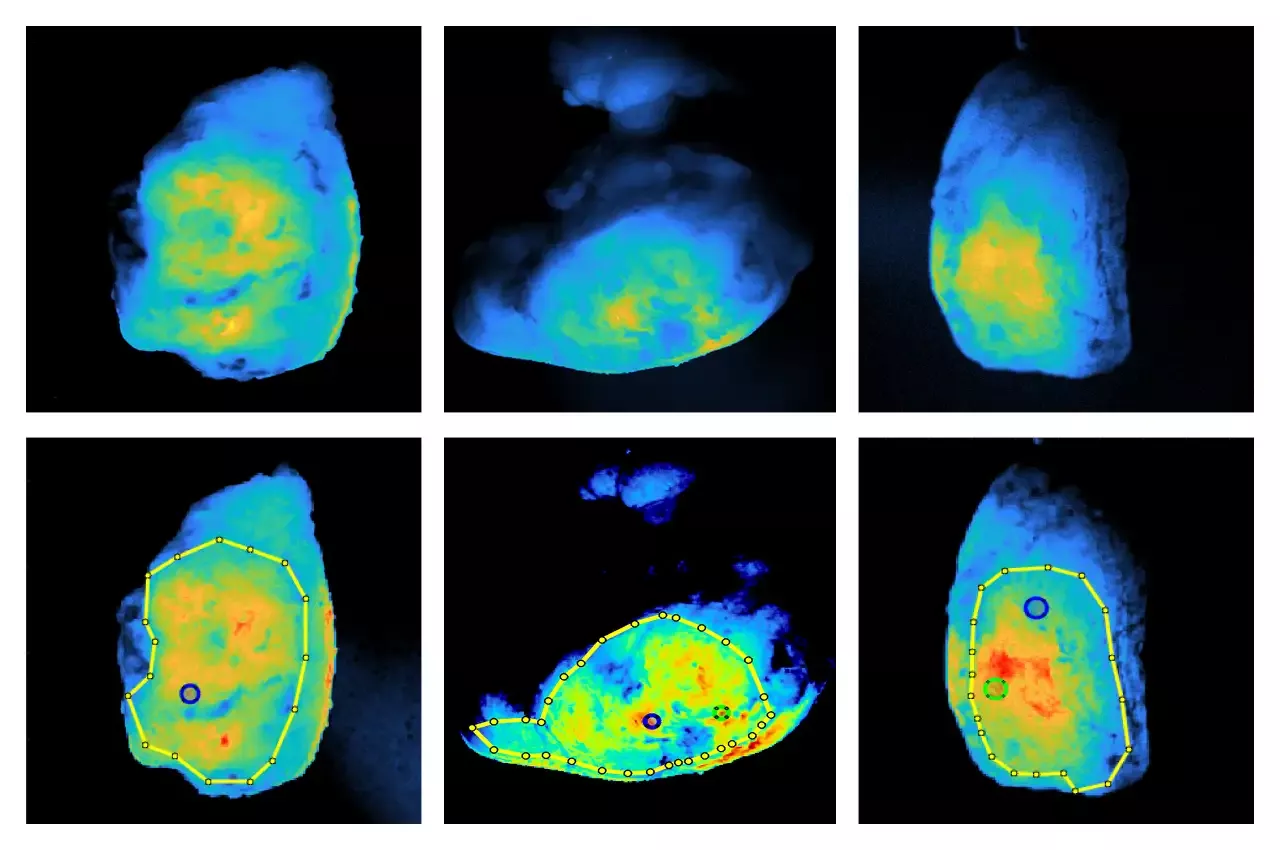
Photo: Scans of tumors using fluorescent molecules that preferentially bind to cancer. Drawn boundaries mark regions of interest defined by surgeons, and the locations indicated mark spots where cancer is closest to the surface (provided).
Disclaimer: “Research reported in this publication was supported by the National Science Foundation under Award Number 1653627. This content is solely the responsibility of the authors and does not necessarily represent the official views of the National Science Foundation.”
Kenneth Tichauer, “CAREER: Development of Enhanced Early Photon Tomography for Cancer Staging,” National Science Foundation; Award Number 1653627




